
George Honos, MD, FRCPC, FACC
Associate Professor of Medicine,
Université de Montréal,
Head of Cardiology
Manager of the CV Program,
Centre Hospitalier de l’Université de Montréal (CHUM)
Simon Kouz, MD, FRCPC, FESC, FACC
Clinical Professor,
Laval University
Chief of Medicine,
Chief of Cardiology,
Director,
Research Unit in Cardiology,
Centre hospitalier régionale de Lanaudière
Anticoagulant therapy has been used to great effect for decades for the prevention of stroke among patients with atrial fibrillation (AF). In recent years, the therapeutic armamentarium has been strengthened considerably, with the addition of anticoagulants acting through novel pathways.
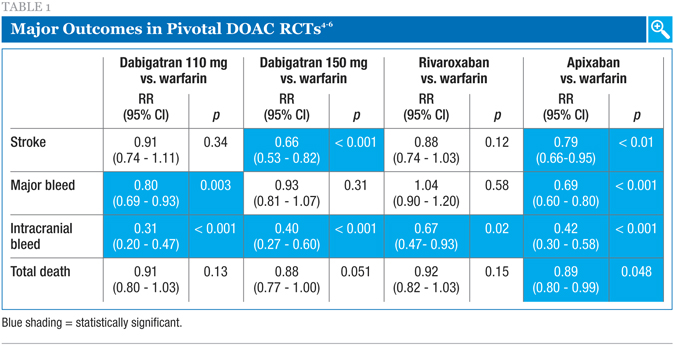
The currently available novel agents are apixaban and rivaroxaban (both oral direct factor Xa inhibitors), and dabigatran (an oral direct thrombin inhibitor).1-3 These direct oral anticoagulants (DOACs) were approved for use on the basis of major clinical trials clearly demonstrating improved risk reductions compared to warfarin for stroke and/or major bleeding events. Table 1 shows the key endpoints of the major randomized, controlled trials (RCTs) conducted with these DOACs.4-6 In these studies, apixaban and dabigatran 150 mg each significantly reduced the risk of stroke, while apixaban and dabigatran 110 mg reduced the risk of major bleeding compared to warfarin. All three novel agents at the doses studied reduced the risk of intracranial (IC) bleeding. Finally, apixaban reduced the risk of death overall.
Extrapolating the results of the DOAC RCTs to all patients is not possible, as the strict design of RCTs yields information that is directly applicable to a relatively narrow spectrum of patients. To control for confounding variables, RCTs restrict enrolment to a pre-specified set of criteria that do not necessarily reflect the profiles of all those who could potentially benefit from these agents.
Research continues using the RCT databases, in an attempt to better identify patient subgroups who do or do not benefit from each of the agents. At the European Society of Cardiology's annual meeting in London in 2015 (ESC 2015, August 29-September 2), there were several presentations and posters providing this type of evidence.
Perhaps more important, as real-world experience with these agents grows, we are beginning to obtain meaningful new information about the DOACs in everyday use. This has involved the study of large databases including patients receiving these medications in clinical situations less stringently defined than in the RCTs. These include purpose-built registries, observational studies, and analyses of healthcare administrative databases. At ESC 2015, a wealth of information was presented using these types of sources. In many cases, these new data reinforce the key learnings from the RCTs: that these agents are efficacious for the reduction of stroke risk among patients with AF; that they are not free from risks; and that there appear to be clinically meaningful differences between the three currently available agents.
The following report provides highlights of registry and other post-marketing data presented at ESC 2015. This is followed by a discussion of the implications of these findings, including how they complement the RCT data and how they may impact clinical practice in Canada.
ESC 2015: New Insights from Real-world Registry and
Other Post-marketing Studies
Bleeding risks with DOACs in the real world (analysis of U.S. claims databases). One of the more compelling datasets presented at ESC 2015 involved a real-world comparison of bleeding risks among patients with non-valvular AF, treated with DOACs.7 The data analyzed in this study came from a large U.S. healthcare database (MarketScan Earlyview insurance claims). The investigators identified more than 60,000 adult patients (aged ≥ 18 years) who received initial anticoagulant therapy with a DOAC or who were switched from warfarin to a DOAC during 2013 and 2014. This included 8,785 patients prescribed apixaban, 20,963 prescribed dabigatran, and 30,529 prescribed rivaroxaban.
For this analysis, patients were followed for up to six months or until a bleeding event, discontinuation/switch of medication or disenrollment from the study. The major endpoints studied were major bleeding and clinically relevant non-major (CRNM) bleeding.
The mean ages of the individuals in the database were similar across the three treatment groups, although the rivaroxaban group did have a significantly lower proportion of patients aged ≥ 75 years (34.5% compared to 38.1% for apixaban and 38.0% for dabigatran). The proportions of patients switched from warfarin in each group were: 17.3% of the apixaban group, 15.7% of the rivaroxaban group, and 4.4% of the dabigatran group. Other differences included a higher proportion of antiplatelet users, greater proportion of comorbidities and slightly higher CHA2DS2VASc and HAS-BLED scores for the apixaban group compared to those taking dabigatran or rivaroxaban.
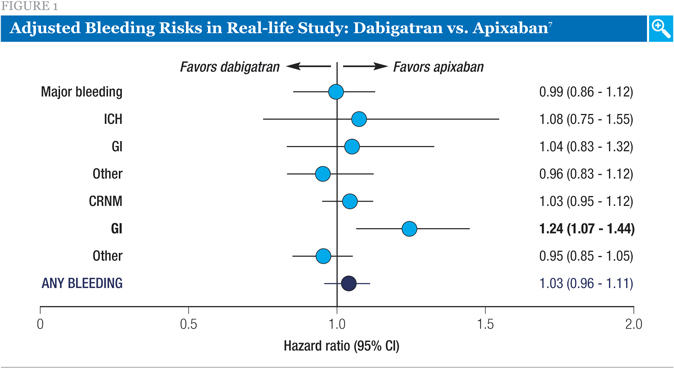
Figure 1 shows the comparison of bleeding risks between the apixaban and dabigatran groups, adjusted for age, sex, comorbidities, and medication use. While most of the endpoints were similar, there was a significantly lower risk of GI bleeding with apixaban compared to dabigatran.
The comparison between apixaban and rivaroxaban is shown in Figure 2. In this analysis, the adjusted risk of each different type of bleeding was significantly higher for rivaroxaban. This included a 34% higher risk of major bleeds (hazard ratio [HR] 1.34, 95% confidence interval [CI] 1.20-1.51) and a 39% higher risk of CRNM (HR 1.39, 95% CI 1.28-1.51).
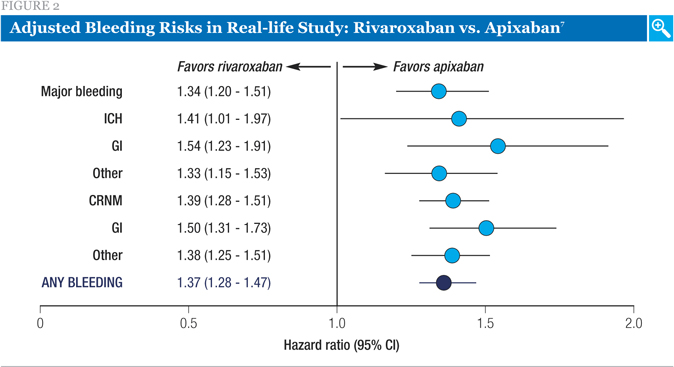
While the investigators acknowledged the limitations of studying claims databases (namely, the potential for missing data and for imprecise timing of treatment initiation, and the fact that there are unmeasured variables which are potential confounders), they concluded that rivaroxaban appears to have an increased risk of major and CRNM bleeding relative to the other DOACs for the first six months after treatment initiation.
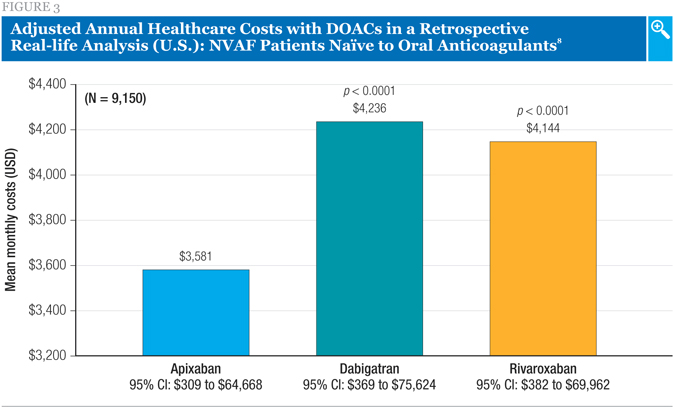
Several other, smaller retrospective analyses involving the same three DOACs were also presented. These included one analysis using data from another U.S. healthcare database, IMS PharMetrics.8 The patients were 9,150 adults with non-valvular AF who were naïve to oral anticoagulant therapy and had newly initiated therapy with a DOAC.8
As was the case with the larger analysis described above, the investigators reported that the risk of major bleeding was significantly increased with rivaroxaban compared to apixaban and that there was no difference between dabigatran and apixaban for this outcome. All-cause hospitalization was found to be significantly higher for rivaroxaban and dabigatran compared to apixaban, with HRs of 1.57 and 1.37, respectively.8 Additionally, all-cause healthcare costs were substantially lower with apixaban compared to either rivaroxaban or dabigatran (Figure 3).8
These pharmacoeconomic findings were echoed by a study from the U.K., which found that apixaban appeared to be a more effective treatment relative to warfarin or dabigatran, at an economically accepted cost.9
Other analyses also showed the same pattern of risk among DOACs in real-world usage, with lower rates of bleeding with apixaban and dabigatran relative to rivaroxaban or warfarin.10-12
Status of — and initial data from — the GARFIELD-AF registry. The GARFIELD-AF registry is an independent academic research initiative involving sites representative of national AF care settings in 35 countries, including Canada.10 At the time of the ESC 2015 presentation, approximately 45,000 patients had been enrolled, out of a target of 57,000.13
The majority of this registry population will be an unselected population of patients prospectively enrolled less than eight weeks after AF diagnosis. In addition to AF, patients have at least one additional investigator-determined risk factor for stroke. There are no entry criteria with respect to pre-existing risk scores. In addition to prospective patients, there will also be a substantial minority (about 5,000 patients) enrolled retrospectively, within six to 24 months of diagnosis. The subjects will be divided into five sequential cohorts by date of diagnosis. Recruitment is expected to be complete by summer 2016, with final analyses planned for summer 2018. Depending on year of a given patient's enrollment, planned follow-up is for a minimum of two and maximum of eight years.
To date, evaluable baseline data are available for approximately 39,670 patients, one-year outcome data for 28,624 patients, and two-year outcome data for 17,162 patients.10
GARFIELD-AF has provided data regarding the increasing use of DOACs over time. In the first cohort of the study (diagnosed in 2010-2011), 4.2% of patients were prescribed a DOAC. This has grown progressively over the subsequent cohorts, with cohort 4 (2014-2015 diagnoses) having 37.0% of its patients treated with a DOAC at baseline.13 Overall, the use of any type of anticoagulant has also grown, increasing from 57.4% to 71.1% from cohort 1 to cohort 4.
The prospective analyses to be performed with the registry data include the rates of stroke/systemic embolism (SE), major bleeding, and all-cause mortality; the types of stroke/SE and major bleeding events and their severity; causes of death; and determination of variables associated with these events.
The data presented at ESC 2015 include the primary two-year outcomes data for cohorts 1 and 2;13,14 an analysis of regional differences in this global database;15 and the impact of gender,16 comorbidities,16,17 and time in therapeutic range18 on outcomes.
For the two-year analysis of cohorts 1 and 2 (N = 17,162), mean patient age was approximately 70 years, with 38% of the total being ≥ 75 years. Comorbidities were common, with 78.1% of subjects having comorbid hypertension, 22% diabetes, 20.6% congestive heart failure, 15% vascular disease (peripheral artery disease or coronary artery disease with history of acute coronary syndromes), 13% prior stroke, and 10% chronic kidney disease (grade 3 or higher).14
Event rates (stroke, major bleeding, all-cause mortality) were highest in the first year after diagnosis, with lower rates in the second year (Figure 4).13,14 Death was the most common major event recorded in the period of observation, with the rates of stroke and major bleeding considerably lower. Mortality was attributed to cardiovascular (CV) causes in 53% of cases.
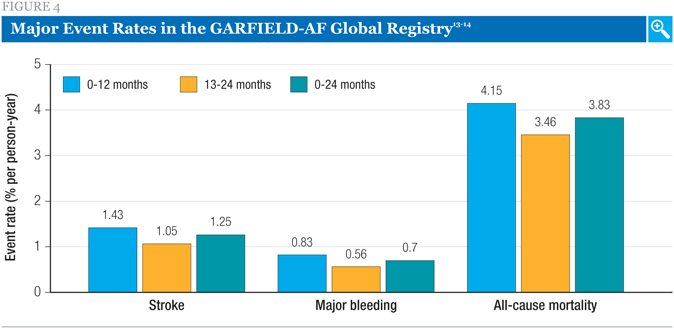
The strokes observed in this data set were primarily ischemic (n = 260). There were also 37 hemorrhagic strokes and 68 strokes of unknown type.
The use of anticoagulant therapy was associated with significant reductions in risk of stroke and all-cause mortality. There was a numerical increase in bleeding with anticoagulant treatment vs. none, but this was not statistically significant. Among the other factors analyzed, age had the strongest association with risk of death, stroke/SE, and major bleeding.14 Higher risk scores (CHA2DS2VASc score or HAS-BLED score) also were clearly associated with increased risks of events.14 The presence of renal disease also was associated with increased risk of the three major event types.14,17,18
With respect to gender, women were 4-5 years older than men at baseline, and a greater proportion of females were aged ≥ 75 years (47% vs. 30% of men).16 Vascular disease was more common among men (17.7% vs. 11.9%), as was history of smoking (50.1% vs. 15.9%).16 After one year, event rates were found to be higher for women than men: stroke/SE rates were 1.62% per person-year for women and 1.17% for men, major bleeding 0.93% vs. 0.79%, and all-cause mortality 4.48% vs. 4.04%.
The effect of anticoagulant therapy on risk reduction also was significantly and substantially greater among men than among women for each of stroke/SE, major bleeding, and all-cause mortality.
Analysis of comorbidities showed that event rates were higher among those with history of chronic kidney disease (CKD) or myocardial infarction (MI), or aged > 80 years. The authors therefore concluded that prescribing anticoagulants should be considered with increased frequency in AF patients with these comorbidities.17
Regarding time in therapeutic range for traditional (vitamin K antagonist, or VKA) anticoagulants, the findings from GARFIELD-AF support those of prior analyses, showing that the time in therapeutic range and frequency in range are both predictive of major outcomes.19
Characteristics and findings from the GLORIA-AF registry. GLORIA-AF is another observational global registry intending to track 56,000 patients, drawn from approximately 1,000 sites in 44 countries.20
Subjects are consecutively enrolled adult patients with newly diagnosed (< three months) non-valvular AF and at least one additional risk factor for stroke (CHA2DS2VASc score ≥ 1).
The objectives are to characterize this real-world patient population and to study patterns, predictors, and outcomes of various antithrombotic treatment regimens for stroke prevention.20
The data available at ESC 2015 were for phase II of the registry, pre-planned for after approval of the first DOAC, dabigatran. Phase I was a cross-sectional analysis conducted prior to approval of any DOACs, and phase III analyses are planned for when baseline characteristics of patients receiving DOACs and VKAs are comparable.20
Among the 15,092 patients recruited in phase II, care settings included university hospitals (32.2%), specialist offices (30%), community hospitals (14.5%), and primary-care practices (11.3%).20
Baseline characteristics were mostly similar to those of GARFIELD-AF: median age 71.0 years, 74.6% with history of hypertension, 24.2% with history of congestive heart failure, 10.5% with previous stroke, and 10.6% with prior MI. Most patients had a CHADS2 risk score of moderate (score = 1; 34.1% of patients) or high (score ≥ 2; 57.8% of patients).
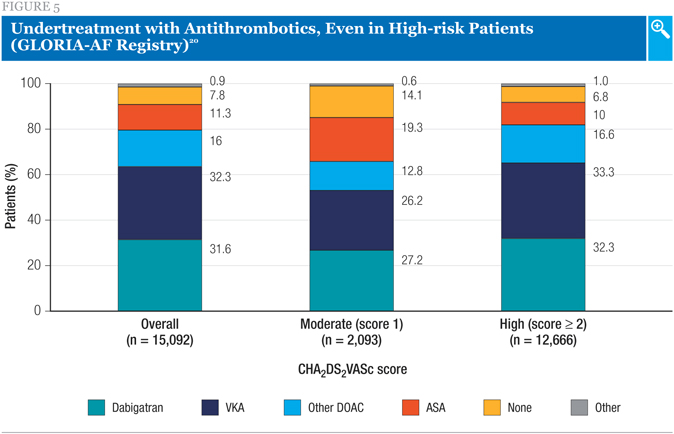
Among the phase II patients, a high proportion was using anticoagulants at baseline, with a greater proportion using a DOAC (47.6%) than a VKA (32.3%; Figure 5). Of note, however, a substantial minority of patients was receiving no anticoagulation, even among the high-risk group.20 A more detailed analysis of this cohort showed that, even among the very-high-risk subgroup of individuals with prior stroke, an antiplatelet alone was the only antithrombotic therapy in 10.8% of patients, while 5.8% received no antithrombotic therapy at all.21 Another analysis showed that, despite possible excess bleeding risk associated with the combination, approximately 5%-7% of patients in this registry received an antiplatelet agent in addition to anticoagulant therapy.22
Findings of the XANTUS study with rivaroxaban. XANTUS was a prospective, observational study that investigated the safety and efficacy of rivaroxaban in routine clinical use for non-valvular AF.23,24 The study included 6,784 patients treated with rivaroxaban at 311 centres in Europe, Israel, and Canada. The mean duration of treatment with rivaroxaban was approximately one year.
The key findings of the study, presented at ESC 2015, were that treatment-emergent major bleeding occurred at a rate of 2.1 events per 100 patient-years, while the overall mortality rate was 1.9 per 100 patient-years. Strokes were less frequent, occurring at a rate of 0.7 events per 100 patient-years.23,24
Analysis of outcomes by adherence with guidelines (EORP-AF registry). The EORP-AF registry is an ongoing European program involving 2,634 patients with AF from nine countries, all cared for by cardiologists.25,26
The goal of the analysis presented at ESC 2015 was to assess outcomes (death, thromboembolism [TE], bleeding, and composite of any TE, CV death, or bleeding) based on adherence to the 2012 ESC guidelines for AF. The investigators classified patients' treatment regimens as being adherent, undertreated, or overtreated relative to the guidelines.
As shown in Figure 6, the rates of TE and the composite endpoint were lower among those considered to be adherent to the guidelines. Significantly better outcomes were observed in terms of mortality and TE, as well as the composite endpoint of CV death, any TE, or bleeding.
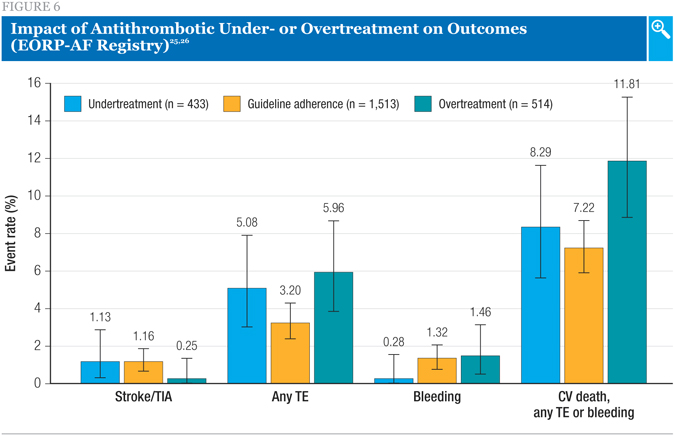
The authors concluded that these results are important, in that the 2012 guidelines advocate an approach in which anticoagulant therapy can be offered to those with at least one stroke risk factor, rather than reserving it for patients with high-risk disease (as was the case with previous guidelines).
Real-world assessment of adherence and persistence to apixaban (AEGEAN study). Another post-marketing study presented at ESC 2015 explored the impact of a patient education program on the rates of adherence and persistence to apixaban therapy.27 This randomized study assigned more than 1,000 patients to receive apixaban and standard care, or apixaban with a structured education program. The program included an additional patient education booklet explaining AF and anticoagulant treatment; reminder tools (key ring, smart message service alert on mobile devices or app); and access to a virtual clinic organized at the country level using staff from existing anticoagulation clinics.
At 24 weeks, adherence and persistence rates were high for both arms (standard of care: 88.5% adherence and 90.5% persistence; education program: 88.3% adherence and 91.1% persistence), with no significant differences between treatment arms. No significant predictors of adherence emerged upon analysis of pre-specified subgroups.
Insights from Randomized, Clinical Trials
Even years after the publication of the RCTs, we still see fascinating results coming from these landmark studies. For example, in sub-analyses of the ARISTOTLE trial comparing apixaban and warfarin, CRNM bleeding was observed in 14.2% of warfarin-treated patients compared to 10.1% among those treated with apixaban (32% decreased risk with apixaban).28
The effect of anticoagulant treatment on renal function was also evaluated in the ARISTOTLE study.29 Overall, the median estimated glomerular filtration rate (eGFR) slightly declined, at a rate of 0.6 mL/min per year. Regardless of eGFR status, apixaban was associated with reduced risk of events compared to warfarin.
Investigators also used the ARISTOTLE data to analyze the impact of apixaban vs. warfarin in patients with no criteria for apixaban dose reduction (from 5 mg to 2.5 mg) and among those with one criterion (age ≥ 80 years, creatinine ≥ 1.5 mg/dL [≥ 133 µmol/L], weight ≤ 60 kg).30 Dose reduction is recommended if a patient has two of these criteria. The investigators reported that the benefit of apixaban 5.0 mg vs. warfarin was similar for those who had one criterion as for those with none, reinforcing the safety and efficacy of this dose among patients with only one criterion.
Data from RCTs were also used to conduct an indirect comparison of stroke and major bleeding outcomes with apixaban (ARISTOTLE study) or rivaroxaban (ROCKET-AF study).31 Using the Matching Adjusted Indirect Comparison (MAIC) method with patient-level data from these studies, there was a significantly lower risk of major bleeds with apixaban vs. rivaroxaban (HR 0.75; 95% CI 0.67-82) and a borderline significant lower risk of stroke/SE (HR 0.88, 95% CI 0.80-1.00).
Data from the ARISTOTLE study were also used to derive a biomarker-based risk score for patients with AF (the ABC-stroke risk score), the validation of which was presented at ESC 2015.32 This new tool was found to perform better than the CHA2DS2VASc score in several populations with AF, and can be recommended for use in clinical practice.
Discussion
The data presented at ESC 2015, particularly with respect to the registry and other post-marketing studies with DOACs, provide interesting complementary information to what we already knew from RCT research.
The comparison of outcomes for each of the DOACs is interesting, as the different definitions of bleeding across RCTs make cross-trial comparisons inappropriate. While registry and other real-world data come along with biases in terms of selection, etc., they can provide clinicians and their patients with reassurance that the RCT findings are supported. Additionally, because of their larger numbers, they could also uncover rare treatment-related adverse events that are not captured in the RCT setting.
However, these kinds of registries are designed to look for any "surprises" that were not captured during RCTs, which themselves do not provide the whole picture. Registries can help confirm observations from trials. At the same time, patients in trials are also real-world patients, and in registries there is no comparator.
In terms of differences between the three DOACs, real-world data support the key RCT findings with respect to bleeding. In RCTs, apixaban and dabigatran have been associated with substantial and significant reductions in bleeding risks relative to warfarin, including major bleeds, whereas rivaroxaban has not been associated with a significant reduction in major bleeds. In the database analyses, apixaban and dabigatran were broadly similar, while there were major and significant differences in favor of apixaban vs. rivaroxaban or warfarin across bleeding categories. This observation is important for clinicians: convenience (e.g., with rivaroxaban) does not necessarily mean that a medication is better or safer. Unfortunately, no data comparing outcomes with the various DOACs in the larger registries (e.g., GARFIELD-AF, GLORIA-AF) were presented at ESC 2015 to lend further weight to these observations. These results are eagerly anticipated.
The observation that apixaban is cost-effective compared to other DOAC options (from two separate sets of authors using different datasets) is also important. These medications are considerably more expensive than warfarin, and to maximize the potential efficacy and safety benefits of getting DOACs to as many appropriate patients as possible, fiscally sound choices will need to be made.
In this regard, it should still be noted that warfarin remains a useful tool in the armamentarium, reducing the risk of stroke relative to no anticoagulant therapy. To be useful, however, one must maintain a time in therapeutic range of 65% or higher.
That many patients are treated with antiplatelet and anticoagulant therapy, observed in baseline data from GARFIELD-AF and GLORIA-AF, is another important observation. The use of antiplatelet agents is recommended for some comorbidities that may exist with AF (e.g., post-MI, peripheral artery disease), while acknowledging the additional bleeding risk associated with such combinations. The decision to initiate or continue multiple concomitant antithrombotics should be made on a patient-by-patient basis by specialists well versed in the patient's comorbidities and overall CV risk.
Several antithrombotic-combination studies currently underway will provide greater clarity on the role of combination anticoagulant and antiplatelet therapies among patient with AF and coronary artery disease (e.g., AUGUSTUS, PIONEER AF-PCI, and RE-DUAL PCI).
Another intriguing topic discussed in this review is the observation that dose reduction of apixaban does not appear to be required for patients with only one risk criterion. Clinicians are well advised to heed these findings, as undertreatment of AF is associated with less favorable outcomes.
Finally, another key observation from ESC 2015 is that even high-risk patients who have had a prior stroke may not be receiving anticoagulant therapy. This, too, points to the need to follow evidence-based guidelines and treat accordingly to optimize the benefits of the currently available therapies.
References
1. Bristol-Myers Squibb Canada. Eliquis Product Monograph. Revised 11 August 2015.
2. Bayer Inc. Xarelto Product Monograph. Revised 20 July 2015.
3. Boehringer Ingelheim Canada Ltd. Pradaxa Product Monograph. Revised 5 June 2015.
4. Connolly SJ, Ezekowitz MD, Yusuf S, et al. Dabigatran vs. warfarin in patients with atrial fibrillation. N Engl J Med 2009; 361:1139-51.
5. Patel MR, Mahaffey KW, Garg J, et al. Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 2011; 365:883-91.
6. Granger CB, Alexander JH, McMurray JJ, et al. Apixaban vs. warfarin in patients with atrial fibrillation. N Engl J Med 2011; 365:981-92.
7. Lip GY, Tepper P, Mardekian J, et al. Real-world comparison of bleeding risks among non-valvular atrial fibrillation patients on apixaban, dabigatran, rivaroxaban: cohorts comprising new initiators and/or switchers from warfarin. Eur Heart J 2015; 36(Abstract Supplement):339; and presented at ESC 2015.
8. Deitelzweig S, Bruno A, Tate N, et al. Major bleeding, hospitalisation rates and healthcare costs among non-valvular atrial fibrillation patients naive to oral anticoagulation and newly treated with novel oral anticoagulants. Eur Heart J 2015; 36(Abstract Supplement):338; and presented at ESC 2015.
9. Lip GY, Lanitis T, Kongnakor T, et al. Comparative cost-effectiveness of oral anticoagulants for stroke prevention in non-valvular atrial fibrillation patients in the UK. Eur Heart J 2015; 36(Abstract Supplement):864; and presented via poster at ESC 2015.
10. Lin I, Masseria C, Mardekian J, et al. Real-world bleeding risk among non-valvular atrial fibrillation (NVAF) patients prescribed apixaban, dabigatran, rivaroxaban and warfarin: analysis of electronic health records. Eur Heart J 2015; 36(Abstract Supplement):1084; and presented via poster at ESC 2015.
11. Lip GY, Pan X, Kamble S, et al. Real world comparison of major bleeding risk among non-valvular atrial fibrillation patients newly initiated on apixaban, dabigatran, rivaroxaban or warfarin. Eur Heart J 2015; 36(Abstract Supplement):1085; and presented via poster at ESC 2015.
12. Deitelzweig S, Bruno A, Trocio J, et al. Early assessment of bleeding-related hospital readmissions among nonvalvular atrial fibrillation patients treated with the new oral anticoagulants using an electronic medical record database in the US. Eur Heart J 2015; 36(Abstract Supplement):1083; and presented via poster at ESC 2015.
13. Kakkar A. Global status of GARFIELD-AF. Anticoagulation and atrial fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
14. Bassand J-P. GARFIELD-AF registry: 2-year outcomes data for cohorts 1 and 2. Anticoagulation and Atrial Fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
15. Goto S. Regional differences in baseline characteristics, use of antithrombotic therapy and outcomes in newly diagnosed atrial fibrillation: Results from cohorts 1–3 of GARFIELD-AF. Anticoagulation and Atrial Fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
16. Camm AJ. Gender and outcomes in cohorts 1–3 of GARFIELD-AF. Anticoagulation and Atrial Fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
17. Goldhaber SZ. GARFIELD-AF: Impact of comorbidities on outcomes in cohorts 1–3. Anticoagulation and Atrial Fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
18. Goto S, Atar D, Bassand J-P, et al. Stroke, major bleeding, and mortality in newly diagnosed atrial fibrillation with moderate-to-severe chronic kidney disease: results from GARFIELD-AF. Eur Heart J 2015; 36(Abstract Supplement):987; and presented at ESC 2015.
19. Haas S. Time in therapeutic range and frequency in range with 1-year outcomes in cohorts 1–3 of GARFIELD-AF. Anticoagulation and Atrial Fibrillation (AF): real life data from the GARFIELD-AF Registry. Presented at ESC 2015.
20. Huisman MV. Global registry on long-term oral antithrombotic treatment in patients with atrial fibrillation: final baseline characteristics of GLORIA-AF Phase II. Presented at ESC 2015.
21. Lip GY, Halperin JL, Diener HC, et al. Use of antithrombotic therapy in patients with atrial fibrillation and prior stroke: insights from the global GLORIA-AF registry. Eur Heart J 2015; 36(Abstract Supplement):243; and presented via poster at ESC 2015.
22. Lip GY, Halperin J, Diener HC, et al. Use of oral anticoagulants in combination with antiplatelet therapy: insights from the GLORIA-AF registry. Eur Heart J 2015; 36(Abstract Supplement):235; and presented via poster at ESC 2015.
23. Camm AJ, Amarenco P, Haas S, et al. XANTUS: Low rates of bleeding and stroke in a real-world prospective, observational study of patients treated with rivaroxaban for stroke prevention in atrial fibrillation. Part of the Session: Registry III - Prevention. Presented at ESC 2015.
24. Camm AJ, Amarenco P, Haas S, et al. XANTUS: a real-world, prospective, observational study of patients treated with rivaroxaban for stroke prevention in atrial fibrillation. Eur Heart J 2015; epub ahead of print, September 15.
25. Lip G. Improved outcomes with ESC guideline-adherent antithrombotic treatment in high-risk patients with atrial fibrillation. A report from the EORP-AF General Pilot Registry. Presented at ESC 2015.
26. Lip GY, Laroche C, Popescu MI, et al. Improved outcomes with European Society of Cardiology guideline-adherent antithrombotic treatment in high-risk patients with atrial fibrillation: a report from the EORP-AF General Pilot Registry. Europace 2015; [epub ahead of print].
27. Montalescot G, Brotons C, Sosyns B, et al. Assessment of an education and guidance program for apixaban adherence in non-valvular atrial fibrillation: the randomised AEGEAN study. Presented at ESC 2015.
28. Bahit MC, Lopes RD, Wojdyla DM, et al. Less non-major bleeding with apixaban versus warfarin among patients with atrial fibrillation: insights from the ARISTOTLE trial. Eur Heart J 2015; 36(Abstract Supplement):338-9; and presented at ESC 2015.
29. Hijazi Z, Hohnloser SH, Andersson U, et al. Efficacy and safety of apixaban compared with warfarin in relation to renal function over time in patients with atrial fibrillation: Insights from the ARISTOTLE trial. Eur Heart J 2015; 36(Abstract Supplement):339; and presented at ESC 2015.
30. Alexander J, Andersson U, Lopes RD, et al. Stroke and bleeding outcomes with apixaban versus warfarin in patients with high creatinine, low body weight or high age receiving standard dose apixaban for stroke prevention in atrial fibrillation. Eur Heart J 2015; 36(Abstract Supplement):345; and presented via poster at ESC 2015.
31. Ishak KJ, Phatak H, Rael M, et al. A simulated head to head comparison of stroke and major bleeding with apixaban versus rivaroxaban in high-risk NVAF Patients. Eur Heart J 2015; 36(Abstract Supplement):743-4; and presented via poster at ESC 2015.
32. Hijazi Z, Lindback J, Ostlund O, et al. External validation of the biomarker-based ABC-stroke risk score for atrial fibrillation. Eur Heart J 2015; 36(Abstract Supplement):710-1; and presented via poster at ESC 2015.
Development of this article was made possible through the support of the Bristol-Myers Squibb/Pfizer Alliance. The authors had complete editorial independence in the development of this article and are responsible for its accuracy. The sponsor exerted no influence in the selection of the content or material published.